Introduction
The largest group of plant pathogens belongs to the Kingdom Fungi, a group of organisms estimated to consist of 1.5 million to millions of species (6,27). Communication about fungal plant pathogens is primarily through the use of scientific names that appear in journals and books where the research of plant pathologists is published. These names encapsulate taxonomic (and recently phylogenetic) knowledge, and keeping the distinction between nomenclature (the formal naming of taxa) and taxonomy (the science of classifying organisms) is fraught with challenges. Our human attempts to create coherent nomenclatural units that are unambiguous and universal for taxa that are variable and evolving will run inexorably into inherent logical and semantic problems (31). An important point to remember is that the purpose of a name is to act as a symbol for communication. Scientific names are abstract concepts that help us to talk about the diversity we encounter in the natural world, used to make reliable generalizations about groups of related organisms, and for information storage and retrieval. Names are excellent tools, but as we continue to refine and revise their application, it is essential to remember Hey’s observations that, "we tend not to notice that our categories lie largely within us ..." and that, "... utility, and not ontological standing, is the key to our appreciation of higher taxa" (31). Modern taxonomists now include many biochemical, ecological, physiological, and molecular characters to delineate new species and genera, which provides more insight into evolutionary history, but often requires a reassessment of nomenclature. Despite the desire for stability, disagreements about species and genus delimitation will persist. This means that scientific names will continue to change, and frustration by the users of names will also continue.
Historically, rules that govern how fungi are named are provided in the international codes of botanical nomenclature, which are revised at each International Botanical Congress (held every six years). Recently, four major changes in the rules that affect the naming of fungi were made at the XVIII International Botanical Congress held in Melbourne, Australia, in 2011, and have since been published as part of the International Code of Nomenclature (ICN) for algae, fungi, and plants, also known as the Melbourne Code (48). The online version of this code is available at
www.iapt-taxon.org/nomen. The Melbourne Code has significant implications for the scientific names of fungi especially those that cause plant diseases and therefore are of concern to plant pathologists. In this article, we introduce the major changes in fungal nomenclature. We also explain the impacts of these changes in fungal names by giving examples of plant-associated fungi. Finally, we discuss possible approaches on how to deal with fungal names in this transitional period.
Major Changes in Melbourne Code
(i) Starting on January 1, 2012, electronic publication is permitted of new scientific names in Portable Document Format (PDF) in online publications with an International Standard Serial Number (ISSN) or International Standard Book Number (ISBN) (Art. 29-31).
It is no longer necessary for new names to appear in printed matter in order to be effectively published. However, publication of names at public meetings, in collections or gardens open to the public, or by the issue of microfilm made from manuscripts is not considered effective publication (48).
(ii) Starting on January 1, 2012, English may be used as an alternative to Latin for the descriptions or diagnoses of new taxa (Art. 39).
Changes (i) and (ii) apply to plants and algae in addition to fungi (48).
(iii) Starting on January 1, 2013, all new fungal names, including new taxa, new combinations, names at new ranks, and replacement names, must have an identifier issued by a recognized repository (Art. 42).
This new requirement took effect on January 1, 2013, after which new fungal names published without an identifier from a recognized repository are not considered to be validly published. In the past ten years, an increasing number of mycologists have been using MycoBank (www.mycobank.org) to register new fungal names. A unique number (e.g. MB 802972) is issued by MycoBank for each registered fungal name, which serves as an identifier to be cited in the publication where the name is proposed. Recently it was decided that three repositories may serve as official repositories of fungal names, i.e., MycoBank, Fungal Names (fungalinfo.im.ac.cn/fungalname/fungalname.html) and Index Fungorum (www.indexfungorum.org) (59).
(iv) Starting on January 1, 2013, the dual naming system for fungi is replaced with one scientific name for each species based on priority.
In previous International Codes of Botanical Nomenclature, Article 59 permitted non-lichen forming fungi to apply different names to different states (pleomorphic fungi). The Sydney Code approved in 1981 introduced specialized terms for the different states of fungi, i.e., the asexual state (anamorph) and sexual state (teleomorph) with the complete fungus and all of its states referred to as the holomorph. The correct way to refer to the holomorph was to use the teleomorph name (if available) preferentially (28). These practices for fungi are now discontinued. According to the Melbourne Code, all legitimate fungal names published prior to January 1, 2013 compete equally for priority, and the sole correct name is now the earliest legitimate name, regardless of the life history state of the type (48). However, in order to maintain nomenclatural stability in pleomorphic fungi, Article 57.2 states that "... in cases where, prior to 1 January 2013, both ... names were widely used for a taxon, an asexual state-typified name that has priority is not to displace the sexual name(s) unless and until a proposal to reject the former ... or to deal with the latter ... has been submitted and rejected" (48).
Changing to One Scientific Name for Each Species of Fungus
Moving to one scientific name for each species of fungus aligns the fungi with the other groups of organisms governed by codes of nomenclature including the International Code of Zoological Nomenclature (ICZN) and the International Code of Nomenclature of Bacteria. For no other group of organisms has two scientific names for one species been allowed except for the fossils and with the Melbourne Code that changed as well. Also in agreement with the other groups of organisms, the correct scientific name for fungi is based on priority, meaning that the first scientific name to be applied to a species is the name that should be used. The Melbourne Code covers algae, plants and all groups traditionally treated as fungi, i.e. the Kingdom Fungi, straminopiles such as the Peronosporales, and the Myxomycetes. Despite their relationship to the true Fungi, it was decided that the Microsporidia should continue to be governed by the ICZN (48).
Integrating the scientific names of fungi into one system allows all entities with clear phylogenetic relatedness within a genus to be called by the same generic name. Previously separate genera were used for the sexual and asexual states of a species. For example, the sexual state species
Cochliobolus miyabeanus
www.google.co.za/search?hl=en&q=Cochliobolus+miyabeanus produces an asexual state, called
Bipolaris oryzae. To those not familiar with the taxonomic history of these fungi, it is not obvious that these names apply to the same species, nor is it apparent that all species described in
Cochliobolus have an asexual state in
Bipolaris or
Curvularia (Fig. 1). As molecular phylogenetic studies increasingly reveal clear relationships among fungi, the use of different generic names for closely related i.e. congeneric species causes confusion. In some cases it results in the redundant description of new generic and species names when the relationship to named genera and species is already known. For example, when an asexually reproducing species with no known sexual state was discovered and determined to belong phylogenetically in the sexual state genus
Chrysoporthe, the previous nomenclatural code (Vienna Code) required that a separate genus be described for this species even though its relationship to the sexual state genus was known (26). Thus the genus
Chrysoporthella was established for one species clearly related to
Chrysoporthe but lacking a known sexual state. Under the Melbourne Code, one can now describe an asexual species in a sexual state genus and vice versa thus eliminating the need, for example, for the genus
Chrysoporthella.
| |

Fig. 1. Phylogram generated from the parsimony analysis based on combined genes of rDNA ITS and GPDH sequence data. Bootstrap values greater than 60 are shown. CBS 730.96 is the ex-epitype of
Curvularia lunata that represents the generic type of
Curvularia. The sequences of
Bipolaris maydis C5 (141-2) were obtained from Berbee et al. 1999. The tree is rooted with
Alternaria alternata EGS 34.0160. | |
For some groups of plant-associated fungi, there will essentially be no changes in scientific names because they do not have pleomorphic life cycles, or the alternative states were not named. These include the straminopiles, Blastocladiomycota, Chytridiomycota, Mucoromycotina, Entomophthoromycotina, Glomeromycota, and most members of the Basidiomycota with the exception of some rust fungi and
Rhizoctonia relatives. Even among Ascomycota there are groups in which the scientific names will remain unchanged as for Taphrinomycotina, most of Pezizales, and even some of the pleomorphic ascomycetes. For example, the generic name
Alternaria and most species in it were described long before any of the sexual state genera or species were established, thus the scientific names in
Alternaria will remain unchanged (71).
Anisogramma anomala, the eastern filbert blight pathogen, and
Cryphonectria parasitica, the chestnut blight fungus, will not change names.
In the process of adopting one scientific name for each species of fungus, it is inevitable that some names in common use will have to be changed. Often specialists in different disciplines prefer different names for the same species. For example, geneticists refer to the causal agent of southern corn leaf blight by the name derived from the sexual state,
Cochliobolus heterostrophus. Under previous codes of nomenclature this name was the correct name to refer to the complete species (holomorph). Plant pathologists who frequently encounter only the asexual state preferentially continue to prefer
Bipolaris maydis. Although considerable disruption will result from changing scientific names for a familiar organism, the end result will be improved communication and a better appreciation for the whole fungus no matter how it manifests itself. In addition, these changes are already stimulating the broadening of research into fungi with little genetic and phylogenetic data, resulting in a clarification of evolutionary relationships. It is our fervent belief that, although this period of transition will be challenging, the end goal of a more stable and simplified naming system will benefit all fungal researchers working in various disciplines.
Following are examples of important plant pathogenic fungi for which integration of scientific names has been initiated. These represent different scenarios and demonstrate the range of issues that are being encountered in fulfilling this initiative.
Well-known genera and species with lists of accepted scientific names forthcoming:
Aspergillus,
Penicillium and
Talaromyces
Aspergillus,
Penicillium and
Talaromyces (Eurotiomycetes, Eurotiales) are well-known mould genera in plant pathology, with hundreds of species in each genus, including many important plant pathogens, mycotoxin producers, industrial fungi, and human and animal pathogens. The International Commission on
Penicillium and
Aspergillus, a commission of the International Union of Microbiological Sciences, has sponsored a series of workshops and associated proceedings (63,64,65), culminating in a formal publication of a list of names in current use (54). The names
Aspergillus and
Penicillium both predate the names of the multiple sexual state genera associated with each group.
Aspergillus is important because of its age and because of the role that one of its species played in the origins of dual nomenclature, and has been the focus of several books [e.g., (2,25,45)].
Aspergillus was first described by Micheli in 1729 (49), making it one of the oldest named fungal genera. The distinctive morphology of the aspergillum (the asexual spore head) remains the principal diagnostic character. By 1854, De Bary (18) noticed that an
A. glaucus mycelium produced sexual spores in a cleistothecium, which had been observed before and given its own name,
Eurotium herborarium (Fig. 2). When De Bary realized that the two forms were different reproductive phases of the same organism,
Aspergillus became the poster child for the nomenclatural predicament that has bedeviled mycologists for over 150 years. Subsequently, many other species named in
Aspergillus for their asexual states were connected ("linked") to a sexual state with a different genus and species name. Eleven sexual genera, with distinct morphologies and ecologies, are known (Table 1). The proliferation of different sexual state names for a genus that most biologists know as
Aspergillus causes considerable bewilderment outside taxonomic circles, interfering with efficient information retrieval (1). Currently there are approximately 250 named species of
Aspergillus (23), many documented in the series of monographs based on cultural and morphological characters by Thom & Church in 1926 (74), Thom & Raper in 1954 (75) and Raper & Fennell in 1965 (57), and a more recent of multigene driven revisions by Houbraken et al. (35) (66,77).
| |

Fig. 2.
Eurotium herbariorum asci: sexual state of
Aspergillus glaucus. | |
Table 1. Synonyms and important species in
Aspergillus,
Pennicillium, and
Talaromyces.
|
Single generic name |
Alternate state synonyms |
Other synonyms |
Important species |
|
Reference |
|
Aspergillus 1729 |
Chaetosartorya | | | | |
|
Emericella | | Genetic model |
A. nidulans | Pontecorvo, 1956; Martinelli and Kinghorn, 1994 |
|
Eurotium | | Grain and food spoilage |
A. glaucus complex | Bennett and Klich, 2003 |
|
Fennellia | | | | |
|
Hemicarpentales | Koenigstein | | | |
|
Neocarpentales | | | | |
|
Neopetromyces | | Plant pathogens, mycotoxins |
A. flavus and relatives | |
|
Neosartorya | | Human pathogens |
A. fumigatus | Calderone and Cihlar, 2002; Latge and Steinbach, 2008 |
|
Petromyces | | Mycotoxins |
A. ochraceus | Bennett and Klich, 2003 |
|
Sclerocleista | | | | |
|
Warcupiella | | | | |
| |
Phialosimplex | | | |
| |
Polypaecilum | | | |
|
Penicillium |
Eupenicillium | | Mycotoxins | many species | |
| | | Storage pathogens |
P. digitatum and
P. italicum (Citrus), P. expansum (pome fruit), P. hirsutum complex (root crops) | |
|
Talaromyces |
Penicillium subgenus
Biverticillium | | Human pathogen |
T. marneffei | |
| | | Biocontrol of soilborne pathogens |
T. flavus complex | |
Because of the ecological and sexual state diversity, the best approach for developing a single name system for
Aspergillus is controversial. The molecular and genomic data examined to date suggests that each of the former sexual genera is monophyletic, but the larger
Aspergillus clade is also monophyletic, albeit with weak basal structure and with the inclusion of some aberrant asexual genera (Phialosimplex, Polypaecilum) among the basal clades (34,52). In an attempt to maintain broad usage of the oldest available name in
Aspergillus for
all of the species of economic significance, the International Commission on
Penicillium and
Aspergillus voted to maintain a broad generic concept for
Aspergillus (39), with all of the formerly sexual genera considered synonyms. Thus, few name changes are expected in the short term as the single name system is adopted, but how stable it will be in the long term remains to be seen. The economic and historical importance of
Aspergillus makes it likely that this taxon will remain at center stage in future discussions about nomenclature and mycological diversity.
Penicillium (Fig. 3) also has a long taxonomic history, since it was first described in 1809 by Link (43), named for its brush-like spore producing heads. Its historical association with named sexual genera is simpler, with two morphological distinct groups called
Eupenicillium (with hard, sclerotium-like fruiting bodies with cell walls of angular cells) and
Talaromyces (with soft fruiting bodies with an outer layer of hyphae). More than 250 species of
Penicillium are now accepted, presented in series of monographs based on cultural and morphological characters by Thom in 1910 (73), Raper & Thom in 1949 (58), Pitt in 1979 (53) and Ramírez in 1982 (56), and a recent series of molecular revisions of individual sections and series by Samson et al. (67) and Houbraken et al. (36,37). In the sense of Pitt (53),
Penicillium was determined to be polyphyletic (5), and the delimitation of new monophyletic generic concepts resulted in the adoption of the name
Penicillium for the former subgenera
Aspergilloides, Furcatum, and
Penicillium (34). The sexual
Eupenicillium is now considered a synonym of
Penicillium; although the former name was used frequently in taxonomic literature, it was rarely applied in experimental or functional literature. The originally sexual genus
Talaromyces was redefined to include many asexual species classified in
Penicillium subgenus
Biverticillium (68). The impact of these changes for plant pathologists is minimal, but the important human pathogen
Penicillium marneffei is now classified in
Talaromyces. Although the latter name change is unfortunate, a satisfactory single name system for
Penicillium and
Talaromyces is now in place (39).
| |

Fig. 3.
Penicillium expansum, the type species of Penicillium, on an apple. | |
Asexual genus has priority with few name changes:
Colletotrichum 1831 vs.
Glomerella 1903
The asexual genus
Colletotrichum contains many well-known plant pathogens that cause anthracnose and black spot diseases on economic crops and ornamental plants (12,38). Around 30 species of
Colletotrichum have been linked to their sexual morphs assigned to the genus
Glomerella (12). The International Subcommission on the Taxonomy of
Colletotrichum (ISTC) has been established and the inaugural meeting was held on 9 August 2012 in Beijing, China. All the ISTC members present at the meeting supported using the asexual name
Colletotrichum over
Glomerella. This decision was not only due to priority (16,78), but also because
Colletotrichum is a more commonly used name in the applied sciences. Database searching done by the ISTC on 9 August 2012 showed generally 80% greater use of
Colletotrichum over
Glomerella in most mainstream search engines and databases such as Google, Google Scholar, Science Direct, and Scopus, and this trend has been historically consistent. In addition, fewer name changes would result by using
Colletotrichum as the name for species in the genus. Although the genus
Vermicularia 1790 is an earlier name for
Colletotrichum, any move to establish
Vermicularia as the genus name for this group would result in unbearable chaos for taxonomists and plant pathologists (12). The first meeting of the ISTC also reached an agreement that an "accepted list of names" will be provided in the light of recent reviews on this topic, such as Hyde et al. (38) "Colletotrichum – names in current use" and Cannon et al. (12) "Colletotrichum – current status and future directions." However, the criteria for making a "list of rejected names" need more consideration.
Sexual genus has priority with agreement on name changes:
Epichloë 1865 vs.
Neotyphodium 1996
The type species of the genus
Epichloë was initially described by Persoon in 1798 (51) as the sexual species
Sphaeria typhina. In 1849, Fries classified
Sphaeria typhina in the genus
Cordyceps subgenus
Epichloë (22). The Tulasne brothers, in 1865 (76), elevated the subgenus
Epichloë to generic status with
E. typhina as the type species. From 1881 until 1982 the asexual state of the broadly defined
Epichloë typhina was classified by Saccardo as
Sphacelia typhina (62). However, Diehl indicated in 1950 (19) that the conidial fructifications of
Epichloë were significantly different from those of
Claviceps and did not apply the form-genus
Sphacelia to the asexual state of
Epichloë. Morgan-Jones and Gams reclassified this species in 1982 in the genus
Acremonium as
A. typhinum in their section
Albo-lanosa (50). Based on their 1996 phylogenetic analysis, Glenn et al. (24) reclassified species of
Acremonium in section
Albo-lanosa in a new form-genus
Neotyphodium. The type species of the genus
Neotyphodium is
N. coenophialum (24) based on
Acremonium coenophialum (50) and includes
N. typhinum.
In addition to their similarity in morphological and ecological characters, phylogenetic analyses of the sexual
Epichloë species and the asexual
Neotyphodium species show that these fungi form a monophyletic group and suggest that the asexual species with unknown sexual states arose from
Epichloë on multiple occasions (69). The generic name for these systemic grass fungal endophytes should be the older generic name
Epichloë. No controversy is anticipated within the community of biologists working on these fungi as a result of transfer of
Neotyphodium species to
Epichloë. Collaborative efforts are already underway to make new combinations in
Epichloë.
Asexual genus to be proposed for conservation, few name changes:
Bipolaris 1959 vs
Cochliobolus 1934
The genera
Bipolaris and
Cochliobolus are fungi with species that cause diseases of economically important grass crops.
Bipolaris is typified by
B. maydis (basionym
Helminthosporium maydis) while
Cochliobolus is typified by
C. heterostrophus (basionym
Ophiobolus heterostrophus). These type species are synonyms i.e. they are the asexual and sexual states of the same species (Fig. 1). This species is the cause of southern leaf blight of maize, a serious disease that occurs in temperate, warm temperate and tropical regions throughout the world (20,72).
At present 115 names have been included in
Bipolaris while 54 names were described in
Cochliobolus (40). Preliminary molecular phylogenetic studies suggest that some species described in
Bipolaris and
Cochliobolus actually belong in the closely related genus
Curvularia (Fig. 1) (46). Both
Bipolaris and
Curvularia have sexual states that have been placed in
Cochliobolus (Fig. 1). Although the sexual genus
Cochliobolus 1934 is an older name than the asexual genus
Bipolaris 1959, the asexual states are more commonly encountered in nature, thus the name
Bipolaris has been used more frequently than
Cochliobolus. In general the sexual states for these species were discovered and named after the asexual states. Thus, the generic name
Bipolaris may be proposed for conservation or protection. The conservation of
Bipolaris would result in only one name change; all other names of
Bipolaris have priority. However, if priority is followed and
Cochliobolus is retained for this genus, 46 names of
Bipolaris would need to be transferred to
Cochliobolus and seven names of
Bipolaris that have priority would replace names currently in
Cochliobolus.
Three species of this genus, specifically
Cochliobolus heterostrophus (=
Bipolaris maydis),
C. carbonum (=
B. zeicola), and
C. sativus (=
B. sorokiniana), have been studied extensively as model organisms. In most cases the publications concerning their genomics and genetics have referred to these fungi using the name in
Cochliobolus, thus it is regrettable to change these important scientific names. However, two of these three species names would need to be changed even if the generic name
Cochliobolus were used because the oldest epithet is currently placed in
Bipolaris. For example
Cochliobolus sativus based on
Ophiobolus sativus 1929 would be replaced by the older name
Helminthosporium sorokinanum 1890 transferred to
Cochliobolus. Similarly
Cochliobolus carbonum 1959 would be replaced by the older name
Helminthosporium zeicola 1930.
Given the number of name changes required if
Cochliobolus were used and the frequency with which
Bipolaris is employed especially by plant pathologists, it seems advisable to conserve the generic name
Bipolaris over
Cochliobolus and conserve the species name
Bipolaris maydis over
Cochliobolus heterostrophus.
Sexual genus proposed for conservation with few name changes:
Nectria 1849 vs.
Tubercularia 1790
Since it was initially described over one hundred years ago, the sexual genus name
Nectria was used for non-black, fleshy, uniloculate, perithecial ascomycetes. Booth (8) established one group within
Nectria called the
N. cinnabarina group that corresponded to the concept of
Nectria in the narrow sense. This genus was further restricted to only 29 species by Hirooka et al. (33). Of the 1104 names described in
Nectria, most have been placed in other genera, such as
Bionectria, Haematonectria,
Lanatonectria, Leuconectria, Neonectria, and
Sphaerostilbella.
The type species of the genus
Nectria is
N. cinnabarina with its asexual state
Tubercularia vulgaris, a species that is well known as the cause of coral spot of hardwood trees. Similarly the asexual genus
Tubercularia is typified by
T. vulgaris (Fig. 4), the asexual state of
N. cinnabarina. Because these two names apply to the same species that are types of
Nectria and
Tubercularia, these genera are synonyms. Within the genus
Tubercularia includes pale-colored sporodochial fungi with slimy aseptate conidia for which 247 names have been described. Seifert (60) examined the type specimens of many of these names and showed them belong in other genera. This genus has never been monographed.
| |

Fig. 4.
Tubercularia vulgaris on a twig, asexual form of
Nectria cinnabarina. | |
The generic concepts of
Nectria and
Tubercularia have varied considerably over time but are now narrowly defined and still synonyms (32,33). The generic name
Tubercularia was described earlier than
Nectria and thus has priority and should be used when moving to one name for fungi. If the name
Tubercularia were used, most of the 29 names in
Nectria would require transfer to that genus. If the generic name
Nectria were protected against
Tubercularia, only three species would require name changes. In addition to the many fewer name changes, the name
Nectria is more precisely defined and familiar to most plant pathologists. As a result it has been proposed that the genus
Nectria as defined in the restricted sense of Hirooka et al. (33) be protected for use over
Tubercularia.
An account of this and other genera in the Hypocreales developed by the Hypocreales Working Committee of the International Commission for the Taxonomy of Fungi has been published (60).
Sexual genus proposed for conservation over a broadly defined asexual genus:
Neonectria 1917 vs.
Cylindrocarpon 1913
The genus
Cylindrocarpon is based on the type species
C. cylindroides. In the broad sense,
Cylindrocarpon included 143 names for species having elongated, multi-septate conidia with broadly rounded ends. When Booth monographed this genus in 1966 (7), he demonstrated that many of these species have nectria-like sexual states. The sexual states of species of
Cylindrocarpon were placed in the genus
Neonectria by Rossman et al. (61). Using a multigene phylogenetic analysis of species representing the breadth of
Neonectria-Cylindrocarpon, Chaverri et al. (15) demonstrated that several major clades existed within this genus and established new generic names for them. Although several segregate genera were recognized, the type species of
Neonectria, N. ramulariae, and
Cylindrocarpon, C. cylindroides, still belong to the same genus and thus are considered synonyms (13,15). In the strict sense
Neonectria includes the cause of European beech bark disease,
N. coccinea; American beech bark disease,
N. faginata; and hardwood canker disease,
N. ditissima (13). Although a number of important plant pathogenic fungi are included in
Cylindrocarpon, many of these are no longer considered in the restricted genus
Neonectria-Cylindrocarpon. "Cylindrocarpon"
destructans, the cause of many root rot diseases, is now placed in a segregate genus as
Ilyonectria radicicola (basionym:
Nectria radicicola) (10). The concept of the genus
Cylindrocarpon based solely on conidial shape is ill-defined including five genera; many of the plant pathogens previously referred to as
Cylindrocarpon have already been placed in more phylogenetically defined genera. On the other hand the genus
Neonectria is well-circumscribed and includes a number of plant pathogenic species. Based on these arguments, it is recommended that the generic name
Neonectria be protected against
Cylindrocarpon.
Sexual genus not monophyletic, controversy over use of asexual genera:
Pyricularia 1880 not a synonym of
Magnaporthe 1972 and
Nakataea 1939 vs.
Magnaporthe 1972
The pathogens causing rice blast and stem rot of rice have had various scientific names applied to them due to difficulty in resolving both taxonomic and nomenclatural issues associated with these fungi.
In 1877, Cattaneo (14) first recorded stem rot, a new disease of rice in Italy and described the causal fungus as
Sclerotium oryzae based on its sclerotial state. About a century later, Krause and Webster (41) established a new sexual genus
Magnaporthe to accommodate this species as
Magnaporthe salvinii. The genus
Sclerotium belongs in Basidiomycota (79) and therefore is not applicable to this ascomycetous fungus.
Nakataea, the conidial state, and
Magnaporthe are congeneric (41). Under the Melbourne Code, the name for the rice stem rot fungus should be
Nakataea oryzae, which is the combination of the oldest legitimate genus and species epithet.
Recently the rice blast fungus,
Magnaporthe oryzae (synonym
Pyricularia oryzae) (17), was shown to be not congeneric to the type species of
Magnaporthe,
M. salvinii, based on phylogenetic analysis, morphology, and ecological characters (Figs. 5 and 6) (44,80). Therefore, the rice blast fungus does not belong to
Magnaporthe as typified by
M. salvinii and should be placed in another genus. The generic name
Pyricularia has been widely used for the rice blast fungus as
P. oryzae and this species is congeneric with the type species of
Pyricularia, P. grisea, with these two species shown to be distinct (17). Thus, the scientific name of the rice blast fungus could revert to the previously used name
Pyricularia oryzae. Alternatively, the fungal name
Magnaporthe oryzae could be considered for conservation. This is allowed under the Melbourne Code but would require a proposal to conserve the generic name
Magnaporthe with a different type species, namely
M. oryzae. Such a proposal would be published in the journal
Taxon, discussed and voted on by the Nomenclature Committee for Fungi of the International Association of Plant Taxonomy (IAPT), and finally voted on at the next Nomenclature Session of the International Botanical Congress in 2017. This issue is a taxonomic one because the generic names
Magnaporthe and
Pyricularia do not compete for priority, i.e., they are not congeneric.

A | |

B |
Fig. 5. Conidiophore (A) and conidium (B) of
Nakataea oryzae (Magnaporthe salvinii, rice stem rot pathogen). |
| |
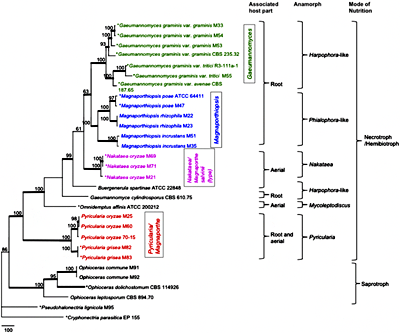
Fig. 6. The most parsimonious tree inferred from the combined SSU, ITS, LSU,
MCM7,
RPB1, and
TEF1 sequence datasets of Magnaporthaceae taxa. MP bootstrap values ≥ 50% are indicated above internodes. Branches in bold have ML bootstrap values ≥ 90% and BI posterior probabilities ≥ 0.95. Mode of nutrition, anamorph (asexual state) and associated host part are mapped on the tree. Type species of the corresponding genera are indicated with asterisks (44). | |
One argument against conserving the name
Magnaporthe with a new type species over
Pyricularia is the excessive number of name changes that would be required for those species currently placed in
Pyricularia. Few name changes would be required if
Pyricularia were accepted. However, 75 names have been placed in
Pyricularia, many of which are plant pathogens of important grass hosts (9). Conserving
Magnaporthe for the rice blast fungus would require changing many names of species currently in
Pyricularia to
Magnaporthe.
Both
Pyricularia and
Magnaporthe are widely used generic names. A discussion and poll are ongoing regarding which name to suggest/reject for the rice blast fungus (http://magnaporthe.blogspot.com). The formal list of accepted/rejected names for the blast fungus and related taxa will be generated by the
Pyricularia/Magnaporthe working group (www.fungaltaxonomy.org/subcommissions) and submitted to the General Committee after the issue is fully discussed in the user community.
How Should Plant Pathologists Cope with the New Fungal Nomenclature?
The change to the nomenclature for fungi will result in the use of some scientific names that are unfamiliar to the user community. In order to minimize this and achieve nomenclatural stability, plant pathologists, mycologists, geneticists, and the broad user community are working together to determine which names to use for pleomorphic fungi. A number of subcommissions of the International Commission on the Taxonomy of Fungi (ICTF) and working groups (www.fungaltaxonomy.org/subcommissions) are putting together lists and proposals about fungal names including "lists of accepted names" and "lists of rejected names." These lists and proposals will be submitted to the Nomenclature Committee for Fungi and General Committee of the IAPT and are subject to the decision of the next International Botanical Congress (29,30). We encourage everyone who works with fungi to get involved in the decision-making process by actively participating in working groups or subcommissions that have been established (www.fungaltaxonomy.org/subcommissions), or to propose a new working group.
As scientific names of plant pathogenic fungi are integrated, lists of accepted names will be published as has been done for well-studied groups such as
Aspergillus and
Penicillium. As the process results in accepted lists, accurate scientific names will be placed on websites such as the USDA-ARS SMML Fungal Nomenclature (nt.ars-grin.gov/fungaldatabases/nomen/nomenclature.cfm) that emphasizes plant-associated fungi, and Index Fungorum (www.indexfungorum.org/Names/Names.asp) and MycoBank
www.mycobank.org that list scientific names of all groups of fungi. These databases will be updated as decisions are made. The National Center for Biotechnology Information (NCBI) manages a taxonomy database (21), which is the standard nomenclature and classification repository for the International Nucleotide Sequence Database Collaboration (INSDC), comprising the GenBank, ENA (EMBL) and DDBJ databases. This widely used resource can be an important aid in transitioning between different Codes. Currently several specialist taxonomists examine new names as they are added to the database, but dual fungal names are still treated inconsistently. An overhaul of fungal names is underway but advice and interaction from the broader fungal research community will be essential.
Acknowledgments
The work is supported in part by the National Science Foundation (grant number DEB 1145174) to N. Zhang. C.L. Schoch was supported in part by the Intramural Research Program of the NIH, National Library of Medicine.
Literature Cited
1. Bennett, J. W. 1985. Taxonomy of the fungi and biology of the Aspergilli. Pages 359-406 in: Biology of Industrial Microorganisms. A. L. Demain and N. Solomon, eds. Addison Wesley, Boston, MA.
2. Bennett, J. W., and Klich, M. A., eds. 1992.
Aspergillus: Biology and Industrial Applications. Butterworths, Stoneham, MA.
3. Bennett, J. W., and Klich, M. 2003. Mycotoxins. Clin. Microbiol. Rev. 16:497-516.
4. Berbee, M., Pirseyedi, M., and Hubbard, S. 1999.
Cochliobolus phylogenetics and the origin of known, highly virulent pathogens, inferred from ITS and glyceraldehyde-3-phosphate dehydrogenase gene sequences. Mycologia 91:964-977.
5. Berbee, M. L., Yoshimura, A., Sugiyama, J., and Taylor, J. W. 1995. Is
Penicillium monophyletic? An evaluation of phylogeny in the family Trichocomaceae from 18S, 5.8S and ITS ribosomal DNA sequence data. Mycologia 87:210-222.
6. Blackwell, M. 2011. The fungi: 1, 2, 3 ... 5.1 million species? Am. J. Bot. 98:426-38.
7. Booth, C. 1966. The genus
Cylindrocarpon. Mycol. Pap. 104:1-56.
8. Booth, C. 1971.
The Genus Fusarium. Commonwealth Mycological Institute, Kew, UK.
9. Bussaban, B., Lumyong, S., Lumyong, P., Seelanan, T., Park, D. C., McKenzie, E. H. C., and Hyde, K. D. 2005. Molecular and morphological characterization of
Pyricularia and allied genera. Mycologia 97:1002-1011.
10. Cabral, A., Rego, C., Nascimento, T., Oliveira, H., Groenewald, J. Z., and Crous, P. W. 2012. Multi-gene analysis and morphology reveal novel
Ilyonectria species associated with black foot disease of grapevines. Fungal Biol. 116:62-80.
11. Calderone, R. A., and Cihlar, R., eds. 2002. Fungal Pathogenesis: Principles and Clinical Applications. Marcel Dekker Inc, New York, NY.
12. Cannon, P. F., Damm, U., Johnston, P. R., and Weir, B. S. 2012.
Colletotrichum: Current status and future directions. Stud. Mycol. 73:181-213
13. Castlebury, L. A, Rossman, A. Y., and Hyten, A. S. 2006. Phylogenetic relationships of
Neonectria/Cylindrocarpon on
Fagus in North America. Can. J. Bot. 84:1417-1433.
14. Cattaneo, A. 1877. Sulla
Sclerotium oryzae, nuovo parassità vegetale, che ha devastato nel corrente anno molto risaje di Lombardia e deI Novarese. Arch. Lab. Bot. Crittog. Univ. Pavia 2-3:75.
15. Chaverri, P., Salgado, C., Hirooka, Y., Rossman, A. Y., Samuels, G. J. 2011. Delimitation of
Neonectria and
Cylindrocarpon (Nectriaceae,
Hypocreales,
Ascomycota) and related genera with
Cylindrocarpon-like anamorphs. Stud. Mycol. 68:57-78.
16. Corda, A. C. I. 1831. Die Pilze Deutschlands. Deutschlands Flora in Abbildungen nach der Natur mit Beschreibungen 3. Abt., tab. 21-32. Nürnberg; Sturm 12:33-64.
17. Couch, B. C., and Kohn, L. M. 2001. A multilocus gene genealogy concordant with host preference indicates segregation of a new species,
Magnaporthe oryzae, from
M. grisea. Mycologia 94:683-693.
18. de Bary, A. 1854. Ueber die Entwickelung und den Zusammenhang von
Aspergillus glaucus und
Eurotium. Botanische Zeitung 12:425.
19. Diehl, W. W. 1950.
Balansia and the Balansiae in America. Agriculture Monograph No. 4, USDA, Washington, DC.
20. Ellis, M. B., and Holiday, P. 1971. Descriptions of Pathogenic Fungi and Bacteria. Nos. 301,302,305-307, 341-349. Commonwealth Mycological Institute, Kew, UK.
21. Federhen, S. 2012. The NCBI Taxonomy database. Nucleic Acids Res. 40(D1):D136-D143.
22. Fries, E. M. 1849. Summa Vegetabilium Scandinaviae Sectio posterior. 259-572:381.
23. Geiser, D. M., Samson, R. A., Varga, J., Rokas, A., and Witiak, S. M. 2008. A review of molecular phylogenetics in
Aspergillus, and prospects for a robust genus-wide phylogeny. Pages 17-32 in:
Aspergillus in the Genomic Era. J. Varga and R. A. Samson, eds. Wageningen Academic Pubs., Wageningen, Gelderland, The Netherlands.
24. Glenn, A. E., Bacon, C. W., Price, R., and Hanlin, R. T. 1996. Molecular phylogeny of
Acremonium and its taxonomic implications. Mycologia 88:369-383.
25. Goldman, G. H., Osmani, S. A., eds. 2008. The Aspergilli. Genomics, Medical Aspects, Biotechnology, and Research Methods. CRC Press, Taylor & Francis, Boca Raton, FL.
26. Gryzenhout, M., Myburg, H., van der Merwe, N. A., Wingfield, B. D., and Wingfield, M. J. 2004.
Chrysoporthe, a new genus to accommodate
Cryphonectria cubensis. Stud. Mycol. 50:119-142.
27. Hawksworth, D. L. 2001. The magnitude of fungal diversity: The 1.5 million species estimate revisited. Mycol. Res. 105:1422-1432.
28. Hawksworth, D. L. 2001. The naming of fungi. Pages 171-192 in: The Mycota, Vol. 7, Part B: Systematics and Evolution. D. J. McLaughlin and E. G. McLaughlin, eds. Springer Verlag, Berlin, Germany.
29. Hawksworth, D. L. 2011. A new dawn for the naming of fungi: impacts of decisions made in Melbourne in July 2011 on the future publication and regulation of fungal names. MycoKeys 1:7-20; IMA Fungus 2:155-162.
30. Hawksworth, D. L. 2012. Managing and coping with names of pleomorphic fungi in a period of transition. IMA Fungus 3:15-24.
31. Hey, J. 2001. Genes, Categories and Species: The Evolutionary and Cognitive Causes of the Species Problem. Oxford Univ. Press, Oxford, UK.
32. Hirooka, Y., Rossman, A. Y., and Chaverri, P. 2011. A morphological and phylogenetic revision of the
Necria cinnabarina species complex. Stud. Mycol. 68:35-56.
33. Hirooka, Y., Rossman, A. Y., Samuels, G. J., Lechat, C., and Chaverri, P. 2012. A monograph of
Allantonectria, Nectria, and
Pleonectria (Nectriaceae, Hypocreales, Ascomycota) and their pycndial, sporodochial, and synnematous anamorphs. Stud. Mycol. 71:1-210.
34. Houbraken, J., and Samson, R. A. 2011. Phylogeny of
Penicillium and the segregation of Trichocomaceae into three families. Stud. Mycol. 70:1-51.
35. Houbraken, J., Due, M., Varga, J., Meijer, M., Frisvad, J. C., and Samson, R. A. 2007. Polyphasic taxonomy of
Aspergillus section
Usti. Stud. Mycol. 59:107-128.
36. Houbraken, J., Frisvad, J. C., and Samson, R. A. 2010. Taxonomy of
Penicillium citrinum and related species. Fungal Div. 44:117-133.
37. Houbraken, J., Frisvad, J. C., and Samson, R. A. 2011. Taxonomy of
Penicillium section
Citrina. Stud. Mycol. 70:53-138.
38. Hyde, K. D., Cai, L., Cannon, P. F., Crouch, J. A., Crous, P. W., Damm, U., Goodwin, P. H., Chen, H., Johnston, P. R., and Jones, E. B. G. 2009.
Colletotrichum-names in current use. Fungal Div. 39:147-182
39.
ICPA (International Commission on
Penicillium and
Aspergillus). 2012. Single name nomenclature in
Penicillium and
Aspergillus. Intl. Union of Microbiological Societies.
40.
Index Fungorum. 2013. Index Fungorum. Online. CAB Intl., Wallingford, UK.
41. Krause, R. A., and Webster, R. K. 1972. The morphology, taxonomy, and sexuality of the rice stem rot fungus,
Magnaporthe salvinii (Leptosphaeria salvinii). Mycologia 64:103-114.
42. Latge, J. P., and Steinbach, W. J., eds. 2008.
Aspergillus fumigatus and
Aspergillosis. ASM Press, Washington, DC.
43. Link, H. F. 1809. Observationes in Ordines plantarum naturales, Dissertatio 1ma (Berlin Ges. NatKde 3: 1-42). Berlin, Germany.
44. Luo, J., and Zhang, N. 2013.
Magnaporthiopsis, a new genus in Magnaporthaceae (Ascomycota). Mycologia. In press.
45. Machida, M., and Gomi, K., eds. 2010.
Aspergillus. Molecular Biology and Genomics. Caister Academic Press, Norfolk, UK.
46. Manamgoda, D. S., Cai, L., McKenzie, E. H. C., Crous, P. W., Madrid, H., Chukeatirote, E., Shivas, R. G., Tan, Y. P., and Hyde, K. D. 2012. A phylogenetic and taxonomic re-evaluation of the
Bipolaris - Cochliobolus - Curvularia Complex. Fungal Div. 56:131-144.
47. Martinelli, S. D., and Kinghorn, J. R. 1994.
Aspergillus: 50 Years On. Elsevier, Amsterdam, The Netherlands.
48. McNeill, J., Barrie, F. R., Buck, W. R., Demoulin, V., Greuter, W., Hawksworth, D. L., Herendeen, P. S., Knapp, S., Marhold, K., Prado, J., Prud'homme Van Reine, W. F., Smith, G. F., Wiersema, J. H., and Turland, N. J., eds. 2012. International Code of Nomenclature for algae, fungi and plants (Melbourne Code) adopted by the Eighteenth International Botanical Congress Melbourne, Australia, July 2011. Regnum Vegetabile 154. Koeltz Scientific Books, Germany.
49. Micheli, P. A. 1729.
Nova Plantarum Genera. 1-234pp.
50. Morgan-Jones, G., and Gams, W. 1982. Notes on Hyphomycetes. XLI. An endophyte of
Festuca arundinacea and the anamorph of
Epichloe tyhina, new taxa in one of the two new sections of
Acremonium. Mycotaxon 15:311-318.
51. Persoon, C. H. 1798. Icones et Descriptiones Fungorum Minus Cognitorum (Leipzig) 1:21.
52. Peterson, S. W. 2008. Phylogenetic analysis of
Aspergillus species using DNA sequences from four loci. Mycologia 100:205-226.
53. Pitt, J. I. 1979. The Genus
Penicillium and its Teleomorphic States
Eupenicillium and
Talaromyces. Academic Press, London, UK.
54. Pitt, J. I., and Samson, R. A. 1993. Species Names in Current Use in the Trichocomaceae (Fungi, Eurotiales). Koeltz Scientific Books, Königstein, Germany.
55. Pontecorvo, G. 1956. The parasexual cycle in fungi. Annu. Rev. Microbiol. 10:393-400.
56. Ramírez, C. 1982. Manual and Atlas of the Penicillia. Elsevier Biomedical Press, Amsterdam, The Netherlands.
57. Raper, K. B., and Fennell, D. I. 1965. The Genus
Aspergillus. Williams & Wilkins, Baltimore, MD.
58. Raper, K. B., and Thom, C. 1949. A Manual of the Penicillia. Williams and Wilkins, Baltimore, MD
59. Redhead, S. A., and Norvell, L. L. 2013. Report of the Nomenclature Committee for Fungi 19: Official repositories for fungal names. Taxon 62:173-174.
60. Rossman, A. Y., Seifert, K., Samuels, G., Minnis, A., Schroers, H.-J., Lombard, L., Crous, P. W., Põldmaa, K., Cannon, P., Summerbell, R. C., Geiser, D. M., Zhuang, W.-Y., Hirooka, Y., Herrera, C., Salgado-Salazar, C., and Chaverri, P. 2013. Genera in the
Bionectriaceae,
Hypocreaceae, and
Nectriaceae (Hypocreales) Proposed for Acceptance or Rejection. IMA Fungus accepted 26 March 2013.
61. Rossman, A. Y., Samuels, G. J., Rogerson, C. T., and Lowen, R. 1999. Genera of
Bionectriaceae,
Hypocreaceae, and
Nectriaceae (Hypocreales, Ascomycetes). Stud. Mycol. 42:1-248.
62. Saccardo, P. A. 1881. Fungi Veneti novi vel critici v. Mycologiae Venetae addendi. Michelia 2:241-301.
63. Samson, R. A., and Pitt, J. I. 1985. Advances in
Penicillium and
Aspergillus Systematics. Plenum Press, New York, NY.
64. Samson, R. A., and Pitt, J. I. 1990. Modern Concepts in
Penicillium and
Aspergillus Classification. Plenum Press, New York, NY.
65. Samson, R. A., and Pitt, J. I. 2000. Integration of Modern Taxonomic Methods for
Penicillium and
Aspergillus Classification. Harwood Academic Pubs., Amsterdam, The Netherlands.
66. Samson, R. A., and Varga, J. 2007.
Aspergillus Systematics in the Genomic Era. CBS Fungal Biodiversity Centre, Utrecht, NL.
67. Samson, R. A., Seifert, K. A., Kuijpers, A. F. A., Houbraken, J. A. M. P, and Frisvad, J. C. 2004. Hylogenetic analysis of
Penicillium subgenus
Penicillium using partial β-tubulin sequences. Stud. Mycol. 49:175-200.
68. Samson, R. A, Yilmaz, N., Houbraken, J., Spierenburg, H., Seifert, K. A., Peterson, S. W., Varga, J., and Frisvad, J. C. 2011. Phylogeny and nomenclature of the genus
Talaromyces and taxa accommodated in
Penicillium subgenus
Biverticillium. Stud. Mycol. 70:159-183.
69. Schardl, C. L. 2010. The Epichloae, symbionts of the grass subfamily Poöideae. Ann. Mo. Bot. Gard. 97:646-665.
60. Seifert, K. A. 1985. A monograph of
Stilbella and some allied Hyphomycetes. Stud. Mycol. 27:1-234.
71. Simmons, E. G. 2007.
Alternaria: An identification manual. CBS Biodivers. Ser. 6:1-775.
72. Tatum, L. A. 1971. The southern corn leaf blight epidemic. Science 171:1113-1116.
73. Thom, C. 1910. Cultural studies of species of
Penicillium. USDA Bureau of Animal Industry Bulletin 118:1-109.
74. Thom, C., and Church, M. 1926. The
Aspergilli. Williams & Wilkins, Baltimore, MD.
75. Thom, C., and Raper, K. B. 1954. A Manual of the
Aspergilli. Williams & Wilkins, Baltimore, MD.
76. Tulasne, L. R., and Tulasne, C. 1865. Nectriei-Phacidiei-Pezizei. Selecta Fungorum Carpologia 3:24.
77. Varga, J., and Samson, R. A. eds. 2008.
Aspergillus in the genomic era. Wageningen Academic Pubs., Wageningen, Gelderland, The Netherlands.
78. von Schrenk, H., and Spaulding, P. 1903. The bitter-rot fungus. Science 17:750-751
79. Xu, Z., Harrington, T. C., Gleason, M. L., and Batzer, J. C. 2010. Phylogenetic placement of plant pathogenic
Sclerotium species among teleomorph genera. Mycologia 102:337-346.
80. Zhang, N., Zhao, S., and Shen, Q. 2011. A six-gene phylogeny reveals the evolution of mode of infection in the rice blast fungus and allied species. Mycologia 103:1267-1276.
